
Titanium porosity treatment is a vital part of making biomedical implants work better. Porous titanium creates a connected three-dimensional structure that works well with the body and helps bones grow. This unique material has become the foundation of bone tissue regeneration engineering. The latest implants now try to copy natural bones in structure, makeup, and flexibility.
Titanium porosity brings great benefits, but surface changes must be done right to get the best results. Studies show chemical treatments work differently – alkali treatment changes the weight by 1.6%, while acid treatment reaches 1.2%, and control groups show 0.8%. On top of that, it needs to match the bone’s elastic strength to stop stress shielding.
Titanium and its alloys need special surface changes to bond well with bone and fight bacteria, which stops implant infections. Each chemical creates different surface patterns, with oxalic acid making the biggest changes to the material. This piece looks at many titanium porosity treatments, how well they work, and where to use them. It helps engineers and doctors pick the right method they need.
Understanding Porosity in Titanium Components
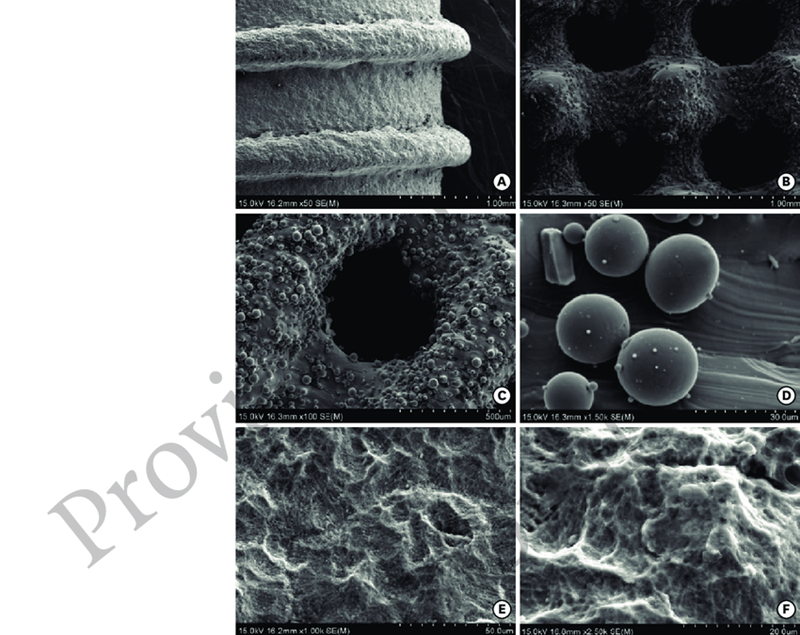
Porosity in titanium components plays a vital role in determining structural integrity and functional performance. Pores change titanium’s properties a lot. This creates materials that work well in medical fields where you need both mechanical strength and biological integration.
Porous Titanium vs Solid Titanium: Structural Differences
The structure and physical properties of porous titanium are different from solid titanium. Solid titanium implants show a high modulus of elasticity (110 GPa) compared to natural cortical bone (7-30 GPa). This creates a big mismatch in stiffness. Porous titanium has an interconnected three-dimensional network of voids that engineers can fine-tune.
Manufacturing processes help control the porosity—the percentage of void volume within the material. Research showed we can make titanium components with porosity from 30% to 75%. This structural change revolutionizes how the material works with biological systems.
Porous titanium can match the human bone’s varied structure. Natural bone has an outer cortical layer and inner cancellous (trabecular) bone with 50-90% porosity. Advanced manufacturing techniques help create this structure in porous titanium. The result is a hierarchical organization with connected pores that look just like natural bone tissue.
Impact of Porosity on Mechanical Strength and Biocompatibility
Mechanical properties and porosity have a clear relationship. Higher porosity leads to lower compressive strength and elastic modulus. Tests show that static compressive strength drops as porosity increases, though not in a straight line. Stress points form at porous areas, making the material more likely to break under load.
The right amount of porosity brings major biological benefits. Studies show:
- High porosity scaffolds (>70%) help bones grow better than low porosity scaffolds (<70%)
- Porosity above 60% works best to form new bone
- Pore sizes between 300-600 μm create the best conditions for bone growth and blood vessel formation
Porous titanium’s elastic modulus can drop to about 5 GPa, matching natural bone closely. This match prevents stress shielding—where stiff implants cause bone loss due to reduced mechanical stimulation. So even with lower strength, porous titanium often works better in load-bearing uses over time.
Porosity turns titanium from a simple support into a living scaffold. The connected pore network helps with key biological processes such as:
- Blood vessel growth and formation
- Bone tissue growing into the implant
- Movement of nutrients and biological fluids
- Cells sticking, moving, and growing
Common Applications of Porous Titanium Implants
Porous titanium’s unique features make it perfect for medical implants. These components work best in orthopedic and dental uses where you need both mechanical support and biological integration.
Orthopedic uses of porous titanium include:
- Hip arthroplasty—about 35,000 hip revision patients in the United States each year use porous titanium implants, especially those with acetabular bone loss
- Spinal fusion devices—3D printed titanium cages with 60% porosity and 500-700 μm pore sizes work better than other materials for fusion quality and stability
- Vertebral disk spacers—designed with changing porosity to match the bone’s natural structure
Dental applications use porous titanium to replace tooth roots and rebuild facial bones. The material promotes osseointegration—where bone connects directly to the implant—making it valuable for these uses.
Porous titanium also works well in custom skull implants, bone plates, and tissue engineering supports. These different uses make the most of titanium’s excellent biocompatibility while solving its mechanical limits through carefully engineered porosity.
Limitations of Traditional Surface Treatments
Traditional surface treatments for titanium have major limitations when used on porous structures. These create unique engineering challenges. Porous titanium implants have a complex architecture that needs specialized approaches beyond the usual techniques. Getting uniform changes throughout connected pore networks remains a tough challenge.
Grit Blasting and Plasma Spraying on Porous Structures
Grit blasting is one of the most common ways to roughen titanium surfaces to improve reactivity and biocompatibility. This technique improves the adhesion strength between substrate and coating for later applications. The process uses various materials as blasting media. Parameters like particle size and blasting pressure directly affect surface properties. Research shows that bigger particles and higher blasting pressure lead to rougher surfaces.
You’ll find several drawbacks when using grit blasting on porous titanium components. The blasting materials—usually alumina (Al₂O₃)—often get stuck in the implant surface. They stay there even after ultrasonic cleaning, acid passivation, and sterilization. These leftover particles can leak into surrounding tissues and interfere with osseointegration. The chemical mix created on the implant surface might also affect titanium’s excellent corrosion resistance in the body.
Plasma spray technology also has its share of problems. While plasma-sprayed titanium coatings have worked well in clinics for decades, their structure causes issues. The process creates open pores, cracks, and areas without coating between splats and particle fragments. This affects the coating’s integrity. The surface roughness becomes harder to maintain as coating layers build up.
These issues get worse with porous titanium structures. Plasma-sprayed coatings show moderate porosity with uneven pore characteristics. They provide simple fixation but lack precise control over important features like pore size, shape, and distribution. The coatings might also peel off—especially where the implant meets the coating—even though they might stick well to bone tissue.
Challenges in Uniform Treatment of Internal Pores
The biggest problem with treating porous titanium structures is getting even changes throughout the porous network. Surface treatments work well on external surfaces but lose effectiveness inside the structure. Many studies show that traditional treatments only work on the surface layer, which means they can’t treat the whole porous matrix evenly.
Chemical treatments face unique challenges with porous structures. Standard chemical etching methods for dense titanium implants use strong acid mixtures. These don’t work well for porous 3D structures because there’s much more surface area exposed to corrosion. You need to fine-tune process parameters carefully to keep macro and micro features while making useful surface changes.
Microarc oxidation (MAO) works great on solid titanium but becomes tricky with porous structures. The coating microstructure gets quite complex. It contains both anatase and rutile forms of titanium oxide mixed with amorphous material. Heat treatment to change these structures is tough because the needed temperature range gets close to or higher than the metal’s melting point.
Porous titanium’s structural complexity makes treatment even harder. These materials often have small pores, uneven pore patterns, poor permeability, or lots of tiny pores in the pore walls. The unique shape of porous implants—including pore size, porosity, and overall structure—makes surface modification much harder than working with nonporous options.
Creating effective surface treatments for porous titanium means balancing opposing effects. Higher porosity helps bone grow better but makes the structure weaker over time. Treatments must also keep the right permeability needed for proper blood flow and bone tissue mineralization.
Acid Treatment for Porosity Reduction

Acid etching is a basic way to change porous titanium surfaces. This method gives exact control over surface roughness and chemistry. Chemical treatments work better than mechanical methods because they reach deeper into porous networks and create unique surface properties that affect biological responses.
H2SO4 and HCl Mixture: Process Parameters
A powerful mixture of sulfuric acid (H₂SO₄) and hydrochloric acid (HCl) changes titanium surface modification. Standard protocols require samples to soak in a 1:1 mixture of 10.6% HCl and 66.3% H₂SO₄ at 60°C for 1 hour with gentle shaking. This ratio works best to etch effectively without damaging the material too much.
Treatment outcomes depend on these key parameters:
- Temperature: Standard procedures need 60°C, though some protocols use room temperature for longer
- Immersion duration: Concentrated mixtures take 1 hour; dilute solutions need 24 hours
- Acid concentration: Stronger acids work faster, but might remove too much material
- Agitation method: Gentle shaking helps acids reach all porous areas evenly
Research shows that tough conditions with higher temperatures and longer soaking times clean surface contaminants like titanium carbide better. Mild etching doesn’t clean surfaces well enough. The acid first breaks through the natural oxide layer, then attacks the titanium underneath.
SEM Analysis of Micro-Scale Roughness
Scanning electron microscopy shows clear changes after acid treatment. The titanium surface develops a distinctive martensitic micro-scale rough structure. This pattern looks nothing like untreated titanium’s smooth surface.
Atomic force microscopy (AFM) measurements prove that surfaces get much rougher after acid treatment. HCl-treated surfaces show the biggest changes, with higher roughness values. SEM images clearly show how acid treatment creates a martensitic micro-scale pore structure on the surface.
Surface analysis reveals key changes in composition. Acid treatment cuts carbon and nitrogen content in half while raising oxygen levels. This means the treatment removes organic contaminants and helps form oxides. X-ray diffraction tests find rutile TiO₂ in acid-treated samples, with the strongest signals coming from samples soaked in mixed acids for 24 hours.
Weight Loss and Mechanical Degradation Post-Treatment
Acid treatment makes samples lose weight. Tests prove that acid-treated samples weigh much less than control groups and alkali-treated pieces. Stronger treatments with higher concentrations, hotter temperatures, and longer times remove more material.
The treatment also weakens mechanical strength. Compression tests show that acid-treated samples aren’t as strong as untreated ones. This weakness can’t be avoided even with the best protocols, which limits what acid-based approaches can do.
Different treatment methods show interesting patterns. Weight changes in simulated body fluid show: alkali treatment (1.6% increase) > acid treatment (1.2% increase) > control group (0.8% increase). Acid treatment makes surfaces better but weakens the structure.
The message is clear – acid treatment must balance better surfaces against weaker structures. Applications needing strong materials should use other surface treatments or gentler acid methods. Yet acid treatment remains useful when biological responses matter more than mechanical strength.
Alkali Treatment and Nano-Scale Surface Modification

Alkali treatment stands out as a better option to modify titanium porosity. It creates nano-scale surface structures and keeps mechanical integrity intact. Biomedical applications have embraced this approach because it maintains both surface bioactivity and structural strength.
NaOH Immersion Protocol for Porous Titanium
Scientists use a standard protocol to treat porous titanium components with sodium hydroxide (NaOH) solution under specific conditions. The process works best when specimens stay in 5M NaOH solution at 60°C for 24 hours with gentle shaking. Here’s how it works:
- Clean with acetone and distilled water (1 hour each)
- Put in 20mL of 5N NaOH solution per disk
- Keep at controlled temperature (60°C) for 24 hours
- Shake gently throughout the process
- Rinse thoroughly with distilled water after treatment
Solution concentration substantially affects treatment outcomes. While researchers have tried concentrations from 0.5M to 18M NaOH, a 5M concentration works best for biomedical applications. Research shows that 240 minutes (4 hours) is the minimum time needed, though 24-hour treatments give the best results.
Formation of Sodium Titanate Layer
The titanium surface changes its topography and composition through a chemical reaction when submerged in alkaline solution. Hydroxide ions first interact with the titanium substrate and create a titanium oxide hydrogel layer. The hydrated titanium oxide then keeps reacting with hydroxide ions to form HTiO3-.
The surface layer that forms has mainly sodium hydrogen titanate (NaxH2-xTiyO2y+1), where 0<x<2 and y=2, 3, or 4. X-ray diffraction analysis shows this layer has both H2Ti2O5•H2O and Na2Ti2O5•H2O compounds. The treatment creates an even nano-scale porous network structure across the surface.
Scanning electron microscopy reveals flake-like nanostructures with pore sizes between 600 and 900 nm. Each flake measures 400-800 nm wide and less than 50 nm thick. This nano-topography makes the surface area much larger, which improves bioactivity by offering many attachment points for biological components.
Preservation of Mechanical Strength
The alkali-treated titanium keeps its strength similar to untreated specimens, unlike acid treatment, which weakens mechanical integrity. Compression tests show no major differences between control and alkali-treated groups. This strength preservation gives alkali treatment a clear advantage over acid-based methods.
Alkali-treated and control groups show minimal weight differences. Acid treatment, however, leads to substantial weight loss and strength reduction. Tests in simulated body fluid show alkali-treated samples gain 1.6% weight compared to 1.2% for acid-treated samples and 0.8% for control groups.
Scientists must control certain parameters carefully. High NaOH concentrations or long heat treatment after alkali immersion might reduce strength. Research shows that using a 1M NaOH solution followed by heat treatment at 600°C for 1 hour reduced the plateau stress of porous titanium by about 25%. This happens mainly due to corrosive damage, which includes grain pull-out and micro-cracks on sample surfaces.
A properly controlled alkali treatment creates a bioactive sodium titanate surface layer while keeping the mechanical properties needed for load-bearing applications intact. This balance between surface modification and structural integrity makes alkali treatment valuable for improving titanium porosity in medical implants.
Simulated Body Fluid (SBF) Testing for Apatite Formation
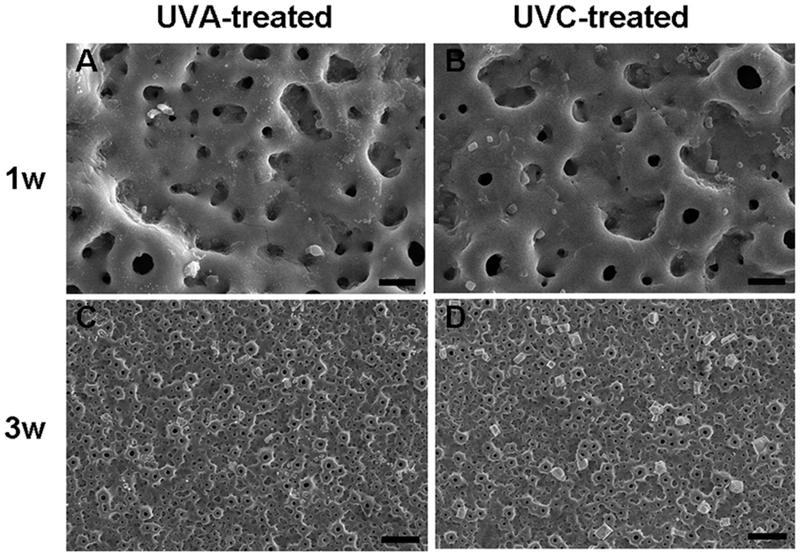
Scientists need standardized testing methods to check how well surface treatments work on porous titanium. Simulated Body Fluid (SBF) immersion stands out as the best way to predict how bioactive materials behave inside the body. This testing method does a great job of copying what happens when titanium surfaces meet body fluids after implantation.
Immersion Protocol and Duration
The standard SBF protocol uses solutions that match human blood plasma’s ion concentrations (Na+ 142.0, K+ 5.0, Mg2+ 1.5, Ca2+ 2.5, Cl- 147.8, HCO3- 4.2, HPO42- 1.0, SO42- 0.5 mM). Scientists clean the samples to remove any surface contaminants before testing. The testing follows these strict rules:
- Scientists place samples in tightly capped polyethylene bottles with 40 mL of SBF per specimen
- The temperature stays at body level (36.5-37°C) during the entire test
- Scientists adjust the solution pH to 7.4 using tris-hydroxymethylaminomethane (TRIS) and hydrochloric acid
- Tests usually run from 1-28 days, though most research focuses on 7-21-day periods
The surface area-to-volume ratio needs special attention for porous titanium. The ISO 23317 standard was created for solid samples and needs adjustments for porous structures because of their much larger effective surface area. Some researchers suggest using fixed mass-to-volume ratios (1.5 mg/mL) to test porous materials instead.
SEM Imaging of Apatite-Like Crystal Structures
SEM reveals clear changes in titanium surfaces after SBF immersion. Apatite starts forming on alkali-treated titanium within hours, and crystal formation becomes visible at 24 hours. Small hemispherical particles appear first and grow in number and size as the immersion continues.
The crystals form in a specific order:
- Sodium ions from the treated surface swap places with H3O+ ions in the fluid to form Ti-OH groups
- Calcium ions join these Ti-OH groups to create amorphous calcium titanate
- The surface layer then takes in phosphate ions to form amorphous calcium phosphate with a low Ca/P ratio (1.40)
- The final step creates bone-like apatite with a Ca/P ratio of 1.65—matching natural bone mineral
Thin-film X-ray diffraction (TF-XRD) analysis shows these structures are hydroxyapatite, with characteristic peaks at 2θ angles of 26° and 32°. SEM observations show that alkali-treated specimens form apatite faster and more evenly than acid-treated or untreated titanium.
Weight Gain Trends Across Treatment Groups
Weight measurements prove that crystals are forming, as more crystal growth means more weight gain. Different surface treatments show clear patterns:
Alkali-treated titanium gains the most weight (1.6%) after SBF immersion, beating both acid-treated (1.2%) and untreated control specimens (0.8%). These numbers show why alkali-treated surfaces work better. Most samples barely change weight in the first few hours, but gain weight quickly once crystals start forming.
Organic compounds can change these results quite a bit. Adding proteins like bovine serum albumin (BSA) to SBF solutions reduces apatite formation because calcium binds with albumin. This finding suggests that standard SBF tests might show more apatite formation than what actually happens in the body, since real body fluids contain organic components.
Changes in solution chemistry back up these weight gain patterns. Solutions with alkali-treated titanium show big drops in calcium concentration, while solutions with untreated specimens stay mostly the same. These changes in ion levels prove that modified titanium surfaces are better at forming apatite, showing that proper surface treatment really does improve bioactive potential.
Quantitative Evaluation of Calcification
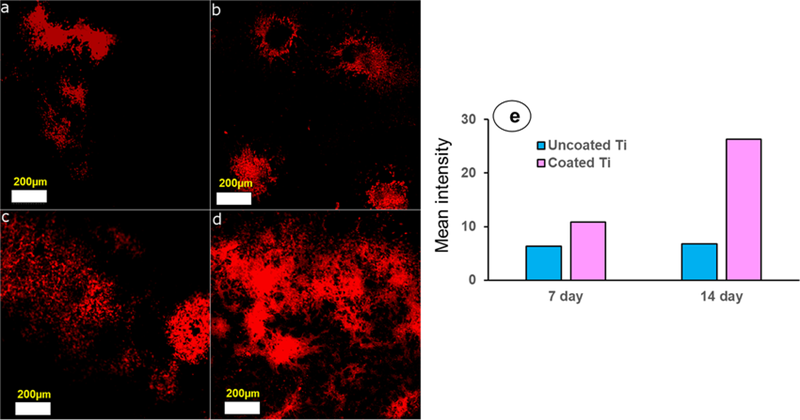
Measuring calcium deposits gives us vital data to match the biological performance of titanium surface treatments. Better measurement methods are changing how researchers look at different porosity treatments for clinical use.
Alizarin Red Staining and Absorbance Measurement
Alizarin Red S (ARS) staining remains the gold standard to measure osteoblast mineralization on titanium surfaces. This method lets researchers analyze calcium-rich deposits that form during osteogenic differentiation. The standard protocol follows these steps:
- Fixation of cell-seeded titanium samples with 4% paraformaldehyde for 15 minutes at room temperature
- Rinsing with distilled water to remove fixative residues
- Staining with 1% Alizarin Red S solution (pH 4.1) for 30 minutes
- Multiple rinses with distilled water to remove unbound dye
Researchers use a standard extraction method to measure results. They start by soaking specimens in 10% acetic acid for 30 minutes to dissolve calcium deposits. The mineralized layer comes off the titanium surface to create a slurry. This mixture heats up to 85°C for 10 minutes and cools on ice for 5 minutes. After spinning at 20,000 × g for 15 minutes, the supernatant is neutralized with 10% ammonium hydroxide. A spectrophotometer measures the solution’s absorbance at 405 nm.
Recent improvements have made detection more sensitive. Adding calcium chloride (1-10 mM) to culture media boosts mineralization based on dose, which makes ARS measurement more precise.
Comparison of Calcium Deposition in Treated Samples
Test results show clear differences in calcium deposits between treated titanium surfaces. Short-term studies (14 days) prove that treated titanium samples have more calcium deposits than untreated ones. Chemical treatments create microstructural changes that affect cell behavior. Surface-treated specimens show many more mineralized nodules by day 14.
Time-based analysis shows calcification follows a pattern, whatever the treatment. The first 14 days show similar calcium deposits across all titanium implants. A clear difference shows up after 21 days. Hydroxyapatite-coated titanium implants have much higher calcium levels.
Culture conditions affect measurement results. Serum-free media leads to higher calcium deposits on hydroxyapatite-coated titanium implants after 28 days. This finding shows why test conditions need standardization to compare treatments fairly.
Titanium implants with existing calcium content, like hydroxyapatite-coated specimens, need baseline calcium values subtracted from test measurements. This adjustment gives a fair comparison between different surface treatments.
Most tests use five samples (n=5) per group. This number gives statistically valid results while keeping the research practical.
Elemental Surface Analysis Using EPMA
Elemental analysis helps us learn about surface composition changes after titanium porosity treatments. Electron Probe Micro Analysis (EPMA) can detect subtle chemical alterations that affect biological performance and corrosion resistance of porous titanium components.
Carbon Contamination Reduction Post-Treatment
Titanium implants face a major challenge with carbon contamination building up on surfaces during ambient storage. This contamination layer disrupts implant osteoconductivity and prevents osteoblast attachment and bone formation. EPMA and similar techniques, like Energy-Dispersive X-ray Spectroscopy (EDS) can measure this contamination by checking carbon content.
Regular titanium shows carbon percentages around 2.60% by weight, creating a barrier between the implant and tissue. Acid and alkali treatments work well to remove this contamination layer. Hydrofluoric acid treatment reduces the C/Ti intensity ratio from 6.3 to 5.3 in 1% HF solutions and drops it to 2.9 in 40% HF solutions. Plasma treatments with argon/oxygen gas mixtures (80%:20%) remove surface carbon even better, with studies showing a 28.1% reduction in carbon content after treatment.
Contamination depth profiles vary significantly between treated and untreated samples. Untreated titanium’s carbon contamination goes deeper into the material. Acid-treated surfaces show much shallower contamination that simple sputtering cycles can remove. Better biocompatibility measurements associate directly with this surface purification.
Oxygen and Sodium Detection in Alkali-Treated Samples
Alkali treatment reshapes titanium’s surface chemistry completely. EPMA shows major compositional changes. Untreated titanium surfaces contain mostly titanium (98%), while alkali-treated specimens show much higher oxygen content (60-68%). Treated samples also contain about 3% sodium, showing clear evidence of sodium titanate formation.
XPS analysis confirms these composition changes. Ti 2p3/2 and Ti 2p1/2 peaks appear at 459.2 eV and 464.9 eV, respectively, indicating Ti⁴⁺ in stoichiometric TiO₂. Alkali-treated surfaces have increased titanium and oxygen content with decreased carbon contamination. A Na1s peak at 1071 eV proves sodium titanate forms during alkali treatment.
These changes go beyond the surface level. EPMA cross-sectional analysis shows sodium and oxygen concentrations decrease gradually with depth, while titanium content rises. Sodium titanate layers usually extend about 1 μm into the material. Heat treatment at higher temperatures (900°C) can push oxygen diffusion depth to around 6 μm.
Mechanical Strength Testing and Statistical Analysis

Mechanical testing serves as the foundation of titanium porosity assessment. This testing helps evaluate different surface treatments through standard protocols and reliable statistical methods.
Compression Strength Measurement Protocol
The testing process needs cylindrical titanium specimens that are 6 mm in diameter and 10 mm in height. Teams test these on universal testing machines. Scientists maintain constant strain rates of 1.0 s-1 in different strain ranges (0.1-0.5). The specimens need graphite lubrication to deform evenly. Temperature readings come from chromel/alumel thermocouples placed in 0.5 mm holes at half-height.
Research shows that compressive yield strength varies a lot between titanium alloys. The tensile-to-compressive yield strength ratios range from 0.79 to 1.24. Scientists track flow stress values and true stress-strain curves. The data show that untreated specimens have much lower flow stress values than heat-treated ones. Some Ti-Al-V alloys reach high compressive yield strengths up to 1276.5 ± 7.3 MPa.
Significance Testing Using ANOVA and Tukey’s Test
The validation starts with Shapiro-Wilk testing to check distribution patterns. Scientists then run parametric tests like one-way Analysis of Variance (ANOVA). ANOVA results often show that radial depth of cut matters most in multi-objective optimization during titanium processing.
Tukey’s Honestly Significant Difference (HSD) test helps analyze variables within treatment groups in detail. Research teams set significance thresholds at α = 0.05 and use SPSS software for analysis. This approach helps identify outliers and compare different treatments reliably.
Conclusion
Titanium porosity treatment plays a crucial role in making biomedical implants better. This piece looks at different ways to modify surfaces, each with its own benefits and limits. The results show that alkali treatment works better than traditional acid-based methods, especially when you need good biocompatibility.
Acid treatments do create useful micro-scale roughness, but they weaken the mechanical structure quite a bit. Alkali treatment, on the other hand, creates a sodium titanate layer with nano-scale surface changes while keeping the mechanical strength intact. This perfect balance makes alkali-treated titanium a great choice for implants that need to bear weight.
Tests with Simulated Body Fluid back up these findings. Alkali-treated titanium consistently forms better apatite. Weight gain patterns across all treatment groups confirm this too – alkali-treated samples show a 1.6% increase compared to 1.2% for acid-treated ones and just 0.8% for untreated controls.
Surface analysis also shows how these treatments cut down carbon contamination and boost oxygen content. These changes in composition directly affect how cells respond and determine the implant’s success in the long run. Scientists can measure these biological responses objectively through techniques like Alizarin Red staining.
Researchers should think about specific application needs when picking titanium surface treatments. Some applications that need maximum mechanical strength might work better with milder modifications, while those focusing on quick osseointegration would benefit from stronger alkali treatments.
Without doubt, these treatment protocols will keep getting better as implant technology advances. The right mix of porosity, surface chemistry, and mechanical strength is the foundation for developing titanium parts that truly match natural bone structure. This deep knowledge of titanium porosity treatment helps engineers and doctors create implants that serve patients better with improved performance and durability.
Key Takeaways
This comprehensive guide reveals critical insights for optimizing titanium porosity treatments in biomedical applications, focusing on surface modification techniques that balance mechanical integrity with biological performance.
• Alkali treatment outperforms acid methods – NaOH immersion creates nano-scale surface modifications while preserving mechanical strength, unlike acid treatments that cause significant structural degradation.
• Porosity enhances biocompatibility but reduces strength – Porous titanium with 60-75% porosity promotes bone ingrowth and matches natural bone elasticity, though compressive strength decreases proportionally.
• Surface treatments dramatically improve apatite formation – Alkali-treated samples show 1.6% weight gain in simulated body fluid versus 0.8% for untreated controls, indicating superior bioactivity.
• Traditional methods fail on porous structures – Grit blasting and plasma spraying cannot uniformly treat internal pore networks, leaving embedded particles that compromise osseointegration.
• Quantitative testing validates treatment effectiveness – Alizarin Red staining and EPMA analysis provide objective measurements of calcium deposition and surface chemistry changes post-treatment.
The key to successful titanium implant design lies in selecting treatment methods that optimize both biological response and mechanical performance. Alkali treatment emerges as the superior choice for applications requiring rapid osseointegration without compromising structural integrity, making it ideal for load-bearing orthopedic and dental implants.
FAQs
Q1. What are the main advantages of alkali treatment for porous titanium implants? Alkali treatment creates a nano-scale surface modification that enhances bioactivity without compromising mechanical strength. It forms a sodium titanate layer that promotes faster and more uniform apatite formation compared to acid treatments or untreated titanium.
Q2. How does porosity affect the properties of titanium implants? Increased porosity in titanium implants enhances biocompatibility and bone ingrowth, while also reducing the elastic modulus to better match natural bone. However, higher porosity also leads to decreased compressive strength, requiring a careful balance for optimal performance.
Q3. What is the significance of simulated body fluid (SBF) testing for titanium implants? SBF testing simulates physiological conditions to assess how titanium surfaces interact with body fluids after implantation. It helps predict in vivo bioactivity and allows researchers to compare the effectiveness of different surface treatments through apatite formation and weight gain measurements.
Q4. Why are traditional surface treatments like grit blasting less effective for porous titanium? Traditional methods like grit blasting and plasma spraying struggle to uniformly treat the internal pore networks of porous titanium structures. They often leave embedded particles or fail to modify deeper layers, potentially compromising the implant’s overall performance and biocompatibility.
Q5. How is the effectiveness of titanium surface treatments quantitatively evaluated? Quantitative evaluation methods include Alizarin Red staining to measure calcium deposition, electron probe microanalysis (EPMA) for surface chemistry changes, and mechanical strength testing using compression protocols. These techniques provide objective data to compare different treatment approaches.

